Following the UK’s top ranking in the recent AMR Preparedness Index, Michael Hodin and Peter Jackson discuss the country’s novel antibiotic subscription model and the key actions it must take to cement its position as a global leader in the fight against AMR.
The UK is poised to lead our world’s response to one of the most dangerous, yet widely overlooked global health challenges that we face: antimicrobial resistance (AMR). According to a recently released Lancet report, AMR claims over 1.27 million lives around the world every year,1 more than HIV and Malaria, and this toll is projected to reach as many as 10 million by 2050.2 The UK’s pioneering AMR policies provide a critical model to avoid these dire projections and galvanise global action.
Like COVID-19 and climate change, AMR is a collective action problem with existential stakes. Our health systems urgently need new antibiotics, but there are no clear incentives to take on the high costs of developing these drugs, which would need to be held in reserve rather than sold and used at scale. Consider that in the UK only one new antibiotic was approved in the 15 years between 1999 and 2014, and fewer than 150 researchers are working on the problem in the industry, predominantly in small-to-medium-sized biotech companies.3
The toll of AMR is already significant and growing. Each year in the UK, AMR causes more than 12,000 deaths4 and incurs over £180 million in costs.5 But these figures pale in comparison to the threat of the decades ahead.
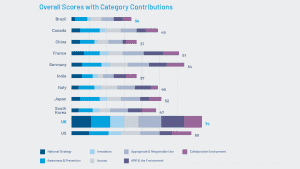
As the challenge worsens, the UK Government and the National Health Service have taken the lead in finding new solutions. The UK was ranked as the top country for proactive AMR policies in the 2021 AMR Preparedness Index, which recognised its comprehensive national AMR strategy and new strategies to encourage innovation.6
The first antibiotic subscription model
The heart of British leadership is a first-of-its-kind antibiotic ‘subscription’ model. Under the pilot programme, launched earlier this month,7 the UK Government pays companies for access to new antibiotics based on their usefulness to the NHS. The initiative, which NHS England and NICE are running, ‘de-links’ the value of new antibiotics from drug sales, reflecting a different kind of valuation focused instead on public health and the value to society of a needed new drug. At the same time, this antibiotic subscription approach limits the over-use of new treatments, preventing future widespread drug resistance from developing.
While global experts have long discussed such a model for AMR, the UK is the first to put this innovative idea into practice. Elsewhere across government, early-stage research and development into new AMR treatments are being supported, for example with £19m of Strength in Places funding for the North-West based Infection Innovation Consortium, iiCON.8 Despite these positive steps, the 2021 AMR Preparedness Index also highlighted a lack of R&D capacity to address the urgent need for new AMR drugs.

The heart of British leadership is a first-of-its-kind ‘subscription’ model for new antibiotics
Four key actions to become a global leader on AMR
As a country, we must now take four key actions to build on our momentum, cement our position as the global leader on AMR, and help defuse this health and economic threat.
First, policymakers can make the antibiotic subscription pilot permanent, increase its funding, and expand its scale across the whole of the UK to spur innovation across the antimicrobial market.
Second, we must implement reforms to reduce the time between regulatory approval and reimbursement decisions. The current lags in this process, which are largely bureaucratic side effects, only serve to impede patient access to urgently needed, new antimicrobials.
Third, we can better coordinate government support, so-called ‘push’ incentives, to help cover the upfront costs of drug development, which are estimated at over $1.5 billion to bring just a single antibiotic to market.9 These initial costs can be especially prohibitive for the small biotech firms and mission-driven research organisations that are primarily focused on AMR. The existing AMR Funders Forum should be beefed up to include industry and private investors to provide more effective and coordinated national leadership on AMR, in the same way, that the industry-led Vaccines Task Force delivered the UK’s world-leading position on COVID-19. By doing this and combining more effective ‘push’ incentives with the ‘pull’ of the antibiotic subscription model, the UK will truly have a best-in-class approach to help innovators overcome financial hurdles and accelerate toward new solutions.
Fourth, the UK can continue to raise the issue, share lessons from our antibiotic subscription pilot programme, and encourage other nations to take similar actions.10 We should build on our global leadership on AMR, fulfilling commitments made during our G7 presidency and helping to foster a more global, action-oriented response.11
While the recent G7 Health Ministers’ Communiqué calling for greater action on AMR is encouraging,12 we know that solving the AMR challenge will require innovative thinking, greater global collaboration, and more decisive action on a broader scale. By expanding our leading-edge AMR efforts at home, the UK can prove that these solutions work and further galvanise the world’s leaders to directly address this mounting crisis.
References
1 https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(21)02724-0/fulltext#seccestitle10
3 https://globalcoalitiononaging.com/wp-content/uploads/2021/06/GCOA-AMR-Preparedness-Index_FINAL.pdf
6 https://globalcoalitiononaging.com/wp-content/uploads/2021/06/GCOA-AMR-Preparedness-Index_FINAL.pdf
7 https://www.england.nhs.uk/2022/06/nhs-lands-breakthrough-in-global-battle-against-superbugs/
9 https://www.nature.com/articles/d41586-020-02884-3, referencing https://pubmed.ncbi.nlm.nih.gov/28888660/
Peter Jackson
Former chairman of the steering committee created to establish the UK’s translational R&D centre focused on antimicrobial resistance, the AMR Centre
Executive Director of Infex Therapeutics
Michael Hodin
CEO
Global Coalition on Aging
This article is from issue 22 of Health Europa Quarterly. Click here to get your free subscription today.

























