
Dr Neda Milevska-Kostova explores how the DRIVE-AMS programme, pioneered by Radboud University Medical Centre, advances antimicrobial stewardship and how changing behaviours and supporting implementation will be essential to tackling antimicrobial resistance.
There is an evident need to address antimicrobial resistance (AMR) globally. In the EU, AMR is estimated to cause around 33,000 deaths every year and accumulates €1.5 billion per year in healthcare costs and productivity losses. According to a recent global burden of bacterial antimicrobial resistance review, the death rate associated with AMR in Europe in 2019 was 67.7 per 100,000 people. An effective response to AMR entails ensuring prudent antimicrobial use, and improvement of surveillance of antimicrobial use, which are both among the key strategic priorities of the EU One Health Action Plan on AMR. This suggests that antimicrobial stewardship (AMS) needs to be further developed and strengthened across the EU.
Antimicrobial stewardship is an established and proven approach to addressing AMR, that can and should be applied in any situation where decision making about antimicrobial therapy is made – at the community level, in nursing homes, outpatient care, and hospital settings. The establishment of an antimicrobial stewardship programme (ASP) in a healthcare facility is a complex process that requires an enabling environment, dedicated resources, knowledge, and processes in place –this is not easily achieved. Even when prerequisites for a functional and effective ASP are in place, and the desired improvements are defined, a challenge remains on how to actually implement and achieve those goals.
Why are current models failing?
In most existing training programmes on the development of ASPs, a lot of emphasis is placed on the ‘WHAT’ of AMS, i.e., the activities and objectives that professionals should perform in daily practice as part of appropriate antimicrobial use such as timely initiation, correct choice, dose, and duration of antimicrobials. Clinicians will be familiar with this as the five rights: right antibiotic for the right patient, right dose, right route and right time (start and duration). But little attention is paid to the ‘HOW’ within an antimicrobial stewardship programme: how to make sure that professionals comply with these WHAT recommendations. Thus, new approaches are needed which require a behavioural change across the entire continuum of antimicrobial use in disease management.
The complexity of AMS and the need for continuous day-to-day improvement demands the establishment of sustainable mechanisms and a culture of collaboration, which takes time and effort to develop and maintain. Training as a single intervention can yield results, but implementation science evidence shows that approaches combining knowledge transfer with other interventions are by far more efficient.
How can we make it work?
Based on this philosophy, in 2018, Radboud University Medical Centre in the Netherlands, under the leadership of Dr Jeroen Schouten, professor Marlies Hulscher, Dr Jaap ten Oever and professor Heiman Wertheim, developed the AMS masterclass and expert consultancy programme (DRIVE-AMS). The aim of the programme is to address the identified disconnect between theoretical knowledge transfer and the capability for implementing effective antimicrobial stewardship interventions as part of an existing or future successful ASP at the facility level. It entails data-driven implementation of a behavioural AMS approach with expert consultancy support for sustainable solutions on appropriate antimicrobial use.
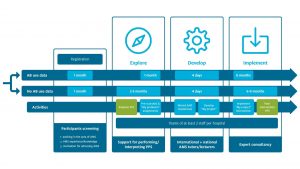
DRIVE-AMS is a six to nine-month programme combining key components of ASP with currently missing elements that can be instrumental in improving the effectiveness of existing and future antimicrobial stewardship interventions. The innovativeness of the DRIVE-AMS approach is three-fold: firstly, the programme is intended for teams (not individuals) from the same facility, which allows for perceiving the AMS from different specialist perspectives and adds value by creating a critical mass within the facility for implementation of a defined AMS intervention. Secondly, the possibility for a tailored approach to create a facility-specific intervention is increasing the chances of process ownership and intervention success and sustainability. Finally, the provided expert consultancy for supporting implementation ensures that mistakes of a learning-by-doing approach are minimised and good practices from other facilities and contexts are successfully incorporated. Through the newly established Specialized Programme for Infectious Disease Care Everywhere (SPICE) expert registry, the pre-trained consultancy coaches are carefully chosen to ensure sufficient expertise in the specific issue being addressed and experience in the given regional and country context.
Another important component is the pre- and post-intervention assessment of antimicrobial use, which in its own right has two main values: on one hand, it allows for an understanding of what needs to be addressed and appropriately sets desired targets for improvement, and on the other, provides evidence for proof of concept of a successfully implemented intervention to be upscaled across the entire facility or to be transposed to other facilities with an interest in excelling in antimicrobial stewardship. This encourages healthcare managers and policymakers to read from the same page, as improved antimicrobial use would inevitably lead to multiple care and economic benefits within a particular healthcare facility and the healthcare ecosystem in general.
To date, more than 100 participants representing nearly 40 AMS teams from facilities in 22 countries have attended, and this year an additional 100-120 participants will experience the programme from different geographic areas including America, Europe, and Africa. Many of these teams have already received, or are currently receiving, expert support for implementation of their developed intervention by Dutch experts during the six months following the AMS masterclass. Both the masterclass and the expert consultancy support have been highly valued by participants who often described them as ‘…probably the most valuable professional experience in the field of antimicrobial stewardship’ that they have had so far, and that it ‘…opens your eyes and shows you alternatives in your real context.’ In other words, for participants, DRIVE-AMS has a unique approach to creating an environment for microsimulation of the process of establishing an effective ASP programme exemplified through an AMS intervention and guided by a team of trainers and expert coaches experienced in the implementation of ASP at the facility level. The programme evaluation and future possibilities were presented in April this year at the 32nd European Congress on Clinical Microbiology and Infectious Diseases (ECCMID) in Lisbon, Portugal.
As this combined skill transfer and implementation process is quite time- and expertise-intensive, in the coming years, DRIVE-AMS and SPICE strategies envisage capacity building of national experts, development of national and regional training and expertise hubs, and development of regional expert registries. To that end, it assumes the continuation of national contextualised courses, extending the competence for measurement and re-measurement of intervention success, building capacity for programme delivery by national experts in national languages, enlisting national and regional expert coaches for providing country-specific implementation support. This new approach of using implementation science for behaviour change, and providing expert support in the implementation of sustainable solutions for appropriate antimicrobial use and multidisciplinary teamwork as indispensable for effective antimicrobial stewardship, can become the norm, and not an exception in mitigating the human-induced AMR tide.
Find out more about the Dutch Antimicrobial
Stewardship programme at
www.radboudumc.nl/en/education/courses/dutch-antimicrobial-stewardship
This article is from issue 22 of Health Europa Quarterly. Click here to get your free subscription today.
























