EU programmes at the Lund University Diabetes Centre, Sweden, provide novel insights into the mechanisms behind the development of cardiovascular disease in diabetes
Cardiovascular disease (CVD) is the major cause of mortality among subjects with diabetes. Diabetes increases the risk of acute myocardial infarction, stroke and peripheral artery disease, as well as the risk of complications after acute CVD events. With an increasing global prevalence of diabetes this represents one of the most important medical challenges worldwide in the years to come.
What is the relationship between atherosclerosis and diabetes?
Diabetes is associated with a more aggressive development of atherosclerosis, which is considered to be the key explanation for CVD in diabetes. Despite being the focus of intense studies, the mechanisms responsible for the aggravated atherosclerosis in diabetes are yet to be fully understood, and most anti-diabetic medications have no or only very limited effect on CVD risk.
Instead, treatment with LDL (or ‘bad’) cholesterol-lowering statins is the most effective way of lowering CVD risk in diabetes, which is somewhat paradoxical because LDL levels are not usually elevated in diabetes.
However, even though most patients with diabetes today receive statins, their risk of cardiovascular death remains twofold higher than the rest of the population. Moreover, the decline in cardiovascular death that has been observed in diabetic patients over the last decades has only paralleled that in subjects without diabetes, suggesting that current therapies target common disease mechanisms but fail to intervene against diabetes-specific disease mechanisms.
It has been assumed that inflammation plays a key role in promoting atherosclerosis in Type 2 diabetes (T2D). This notion makes sense because T2D is generally associated with a low-grade inflammatory state, and it is well known that inflammation promotes both atherosclerotic plaque growth and the destabilisation of plaques that cause acute events.
What role do vascular injury and repair play?
However, recent studies performed as part of the FP7 (Seventh Framework Programme) project Vaccination In Atherosclerosis (VIA) and the Innovative Medicine Initiative project SUrrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools (SUMMIT) at the Lund University Diabetes Centre (LUDC) have questioned this concept and shown that an impaired balance between vascular injury and repair may be more important.
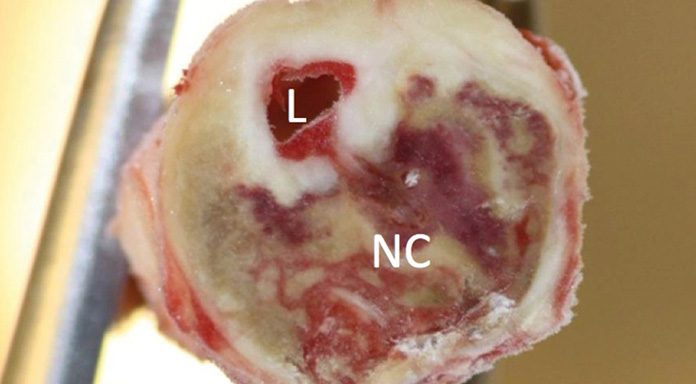
The first indications that this is the case came from studies in which carotid plaques removed at vascular surgery from patients with and without T2D were investigated using a number of morphological and molecular techniques. We unexpectedly found that there were no signs of increased inflammation in plaques from T2D but that these instead contained less of the fibrous proteins collagen and elastin. This suggested that the vasculature in diabetes is subjected either to increased tissue damage or to an impaired capacity to repair such damage – or possibly both.
There is an interesting and perhaps even likely possibility that T2D initially indeed promotes atherosclerosis through fuelling inflammation but that today this becomes less important once statin treatment is initiated as statins are known to reduce plaque inflammation. Consequently, this would then give other disease mechanisms time to gain importance.
How do EPCs interact with diabetes and CVD?
The possibility that impaired tissue repair capacity could contribute to plaque vulnerability in diabetes is of particular relevance in view of the chronic foot ulcers that many patients with diabetes suffer from.
Endothelial progenitor cells (EPCs) derived from the bone marrow play an important role in the formation of the new blood vessels that are needed for the healing of cutaneous wounds. The ability to generate EPCs is reduced in diabetes, which is considered to be one of the most important explanations for the development of chronic foot ulcers. The activation and expansion of EPCs is dependent on stem cell factor (SCF).
There is evidence that EPCs also play a critical role in maintaining general endothelial integrity. As EPCs are present only in very low numbers in the circulation and technically difficult to analyse in large clinical studies, we instead investigated plasma levels of SCF as a surrogate marker for this process.
Using the proximity extension assay (PEA) technology (www.olink.com) we determined circulating SCF in 4,742 middle-aged men and women participating in the prospective population-based Malmö Diet and Cancer Study (MDCS). The baseline blood samples were collected between October 1991 and February 1994 and the incidence of cardiovascular events and death monitored through national registers during a mean follow-up of 19.2 years.
We found that SCF levels were reduced by 15% in subjects with T2D and showed inverse correlation with metabolic risk factors such as fasting glucose and triglycerides in non-diabetics. Moreover, low baseline levels of SCF were associated with higher cardiovascular mortality as well as risk for development of myocardial infarction, stroke and heart failure.
These observations suggest that a reduced capacity of SCF-mediated generation of EPCs may contribute to impaired vascular repair and increased CVD risk in diabetes as well as in non-diabetics with metabolic risk factors.
How is the LUDC working to elucidate our understanding of these mechanisms?
As we reasoned that one possible cause of the reduced content of fibrous proteins in plaques from patients with diabetes could be an enhanced expression of matrix proteinases (MMPs), we went on to focus on circulating matrix metalloproteinase levels.
Using a cohort of 500 subjects with T2D and clinically manifest CVD, 500 subjects with T2D but without CVD matched for age, sex and duration of T2D and 500 matched non-diabetic controls enrolled at four European centres within the framework of SUMMIT, we could demonstrate subjects with T2D have significantly increased plasma levels of MMP-7 and -12. Moreover, plasma levels of both MMPs correlated with the severity of atherosclerosis in T2D subjects and were higher in T2D subjects with prevalent CVD than in those without. We next analysed determined plasma levels of MMP-7 and -12 in MDCS baseline samples and found that high levels were associated with an increased risk for development of myocardial infarction. In a separate study based on 205 carotid endarterectomy samples we could demonstrate that high plasma levels of MMP-7 were associated with a lower plaque collagen and elastin content. These observations argue that diabetes is associated with an increased MMP expression that contributes to plaque destabilisation and risk for acute cardiovascular events.
There is convincing experimental evidence that atherosclerosis develops in response to vascular injury caused by dyslipidemia and other cardiovascular risk factors. However, there has been a lack of clinically useful biomarkers reflecting this injury.
We took advantage of the fact that soluble forms of so-called ‘death receptors’ exist in plasma to investigate if these could be used as markers of cell injury and death. The death receptors constitute a family of cell surface receptors, including Fas, TNF receptor-1 and TRAIL receptor-2, that mediate activation of apoptosis (also referred to as programmed cell death).
We found that activation of Fas results not only in activation of apoptosis but also in the release of all three receptors in soluble forms, suggesting that these indeed represent markers of apoptosis.
We next determined baseline levels of soluble Fas, TNF receptor-1 and TRAIL receptor-2 in the MDCS cohort. The results indicated that the rate of cell death by apoptosis was about 10% higher in subjects with diabetes and also showed that the plasma level of soluble death receptors correlated significantly with most other known cardiovascular risk factors. High levels of soluble death receptors were associated with an increased risk of myocardial infarction, stroke and cardiovascular death.
How will these findings inform future strategies to prevent CVD in diabetes patients?
Taken together these studies are in line with the notion that statins reduce inflammation as a driver of atherosclerosis, allowing other factors involving injury and impaired vascular repair to become increasingly important.
To further improve CVD prevention in diabetes, we propose that novel therapies targeting these processes need to be developed.