
The Addenbrooke’s Air Disinfection Study looks set to shape future IPC guidance concerning the implementation of air filtration systems in healthcare settings.
The Addenbrooke’s Air Disinfection Study (AAirDS) is funded and supported by UK Health and Security and will comprehensively challenge the role aerosol transmission has on a range of hospital-acquired infections. With a world-leading national, multi-disciplinary team this study could shape future infection prevention and control (IPC) guidance around the use of air filtration systems in healthcare.
What is the Addenbrooke’s Air Disinfection Study (AAirDS)?
The AAirDS uses air filtration units (AFUs) in combination with environmental testing via air and surface sampling, historical and prospective infection data, and indoor air quality (IAQ) data to track and monitor aerosol movements within wards. Approximately 2400 patients will be included in the study by the end of the 12-month study period.
The technology deployed is provided by Air Purity Ltd which has been testing and developing within Cambridge University Hospitals NHS Foundation Trust since 2020. The AFU include ‘Aero Titans’ which consist of larger units in the hospital corridors and medium-sized units within the bays. ‘Aero Sentinels’ are also deployed to read the environment in real-time and relay all information back to the units and Trust to ensure they are running correctly and controlling the ward environment.

What will this study tell us about infections in hospitals?
We know that certain infections peak at specific times of the year, for example, RSV in early winter, influenza in mid to late winter, and blood-borne infections in the summer. The environmental conditions that these pathogens are exposed to such as temperature and humidity, coupled with human factors such as the volume of individuals indoors as well as societal behaviour also impact on wider community transmission of pathogens. By sampling the air for viruses, bacteria, and fungi throughout the study and gathering indoor air quality (IAQ) and real-time patient infection data, we will glean insights into the conditions that favour specific pathogens. This may open new avenues for mitigation; for example, if certain pathogens thrive in drier air humidification may be one control strategy.

What can be done now to reduce infections, especially COVID?
There are two main areas to address when dealing with an airborne pathogen as transmissible as SARS-CoV-2, in particular, the BA.1 and BA.2 omicron variants. These are:
- Short-range; and
- Mid to long-range.
Short-range
Wearing higher grade respiratory protection such as FFP2/3 respirators reduces both staff and patient infections. It is not clear how much transmission within hospitals is short-range from patient to patient, but we do know that nine out of every 10 infections caught in hospital come from other patients. Short-range transmission within hospitals will therefore be driven by asymptomatic infected healthcare workers and visitations infecting patients through close contact, which is less affected by ventilation and other mitigations such as AFUs. The other main way of reducing infections is via improved source control; that is the widespread wearing of masks by both patients and staff including those known to have the SARS-CoV-2 infection, in order to lower their emission of infectious viral particles. Upgrading PPE for staff working on COVID-19 wards in Addenbrooke’s Hospital in Cambridge cut hospital-acquired infections dramatically.
Mid to long-range
Secondary transmission to patients and staff can occur at distances further than two metres due to the build-up of infectious particles (aerosols) that accumulate in poorly ventilated environments, such as those seen on hospital wards. This mode of transmission can be significantly reduced with ventilation and air cleaning, either by filtration or disinfection (usually via ultraviolet light) as has been demonstrated by earlier studies at Cambridge University Hospitals.
What are the challenges of air filtration systems in hospitals?
Hospital wards, particularly those forming part of the older NHS estate, are often restricted for space such that any air cleaning intervention must be workable around other pieces of equipment necessary for inpatient care. They also need to be effective at longer distances than most domestic machines and quiet enough to be situated close to patients. AFUs can also create unpleasant draughts for patients so the direction of any air outflows needs to be actively managed.

Should hospitals wait for the results of AAirDS before implementing an air cleaning solution?
Previous studies have already demonstrated the utility of AFUs in managing outbreaks with problematic fungal pathogens. With the airborne transmission of SARS-CoV-2 and other respiratory viruses now accepted by the World Health Organization individual hospital trusts should not wait for the outcome of AAirDS, particularly with nosocomial transmission at record levels. The study is however expected to report in mid-2023 adding vital information with respect to the practicalities and cost-effectiveness of air filtration and environmental disinfection.
How should hospitals implement an air filtration solution?
We have already demonstrated that appropriate commissioning work following the deployment of air filtration units in zones within wards is vital to understanding and actively managing the air in ward environments. Environmental sensors in our study relay data in real-time to show how an environment behaves and if sufficient filtration is taking place to be assured the air is cleaned of pathogens. Without understanding how an AFU is cleaning the air it is challenging to understand the distances with which a single AFU is having an effect. The AFUs used in AAirDS have been shown to have a cleaning effect beyond 15 metres.
We would therefore recommend that as well as focussing AFUs on the poorest ventilated wards, or those with frequent outbreaks, that particular monitoring is employed after installation to quality assure the technology being deployed.
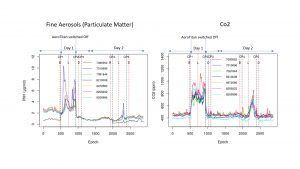
The graphs show a natural experiment as the AFU should not have been switched off (pictured below). Each line corresponding with a number on the graph legend indicates an Aero Sentinel sensor which are placed over half a ward space. The significance is the unit reduces aerosols and CO2 concentrations and shows a clear difference (the build-up) when the unit was switched off.
Aero Titan had a huge impact on the significance of reductions in the two precursors for secondary infections via aerosol dissemination, for a more in-depth look into this scientific publication please see: Impact of supplementary air filtration on airborne particulate matter in a UK hospital ward.


Some tips on implementing an AFU device
- Check space and plugs: plugs are a premium in wards as is space, in some instances careful planning should be made to ensure a placement can be suitable to accommodate the needs of the ward.
- Size of unit (ergonomics): if the unit does not go past your hip, it is very unlikely it will have an effect on the head height air you and so many others share. We see fast rate on travel in fine aerosols at higher heights, much consideration needs to be taken regarding how a unit sits in an environment and at what height.
- Air Flow (CADR) / noise: units ideally need to be as powerful as possible. For example, the Aero Titan 3000 moves approximately 3,700 m3/hr but captures aerosols and moves clean air at multiple height ranges and is very effective on lower speed settings. Test the noise levels of the units; the biggest improvements and adjustments Air Purity had to make were for patient and staff comfort, if a unit is underpowered it will not be noisy but it will also not help in controlling the ward space to reduce secondary infections.
- Air change rate calculation: this rule should not determine an implementation; placement will always be the biggest determining factor. The reality is depending on how the air is cleaned and the direction it flows, where openings are (doors, windows), the volume of people at maximum occupancy, how an HVAC is configured, the volume displacement of people moving, and the temp and humidity, it will determine what the real-world clean rate of a room would be. Typically, units will clean very often within a small space but be drowned out from the larger environment and thus not reach very far at all.
- Evaluate the environment: following on from the previous point a form of measurement before and after is needed, and this needs to be maintained which takes resources and education.
- After service: any suppliers with no after service (teams to maintain the units) has no diligence for a hospital environment. A robust commissioning process, regular planning, and servicing are all needed. Remember this is to prevent and reduce infections and should be regarded as important as medical equipment.
- Lab testing / small room testing: from the most affordable to the most expensive units nearly all will clean the air in a small, controlled space. However, in an open environment, we have seen so many types of units fail, and full outbreaks still occur. The reality is the industry currently lacks guidance in commission and standards for implementation, so it is a complete minefield and determined by the supplier. The AAirDS will help to change this from the results to be published later this year and through 2023, however, please feel free to contact the Cambridge University Hospital team, or Air Purity directly.
Further Guidance
Air Purity provides free assessments and recommendations through mapping aerosol movements at no obligation or cost. Data collected will be shared and remain for UKHSA and Trust use only, staff are FFP3 fit tested and can evaluate Red wards if necessary.
Where can I get further information?
You can visit www.aairds.com or contact Air Purity UK directly for a free no-obligation evaluation of your clinical environment.
This article is from issue 21 of Health Europa Quarterly. Click here to get your free subscription today.

























