
SyrinJector introduces an innovative solution to the enormous challenge in the digital health of livestock farm world.
According to SyrinJector CEO Nahum Cohen: “The current situation in the veterinary mass vaccination market is fraught with a number of major shortcomings, the most important of which is the inability to systematically monitor the vaccination processes carried out.”
SyrinJector’s development is at the forefront of innovation by integrating a smart digital injection patented system and IoT capabilities into a product that is expected to make the animal health world more traceable, efficient and safer for both humans and farm animals.
Cohen founded SyrinJector along with Udi Nechmad. He recalls: “In 2012, a close friend – Udi, who owns livestock syringe company Nechmad Ltd. (www.nechmad.com) – came up to me, an aeronautical engineer, and told me he wants to take syringes to the next level. I loved the idea of creating a syringe gun ‘on steroids’. The very next month we established the company.”
Nechmad recalls: “It took a long process of research and development. We started with several concepts until the right concept appeared.”
The product, which will enter the market in the third quarter of 2019, will allow, for the first time, digital capabilities which include information management, full automation and high precision. SyrinJector offers a safe and smart solution for the benefit of farm animals to the next generation of farmers around the world.
Success potential
Elkana Pressler, the company’s business and market development manager, says: “The vaccine market in the livestock industry is constantly growing.”
According to a Markets and Markets forecast, the market for veterinary vaccines is expected to grow between 2015 and 2020 at an average annual rate (CAGR) of 5.5% – and reach $7.197bn by 2020. SyrinJector’s innovation is expected to help it penetrate this market effectively and grow in it rapidly.
“The success potential of SyrinJector is particularly great thanks to a business model based on partnerships with corporations who distribute veterinary systems and equipment,” continues Pressler.
The end customers for the product are livestock farms and integrations, which are expected to significantly improve the welfare of the livestock and their ability to maintain their health, as well as that of the consumers.
For the pharmaceutical companies, the product will provide tracking capabilities – unique information and vital statistics about how their products are being used – and minimise counterfeits.
Additional revenue streams include:
- Comprehensive database and data analysis for government inspection entities
- Sale of the platform and software licence
- Sale of consumable accessories – cleaning/disinfecting module and software license for non-syringe customers such as authorities
- Sale of services after the end of the warranty period.
Cohen feels fully reassured: “SyrinJector is funded by top veterinarians and business investors from Israel and abroad. I like it when professionals put their smart money on us – it gives us a great backwind.”
Among other things, the company has been selected and funded under the European Innovation Council pilot’s SME Instrument as part of the community of tomorrow’s innovation leaders of the SME Horizon 2020 programme.
The company will launch its beta site in February 2019 and is now signing a preliminary marketing agreement with a major distributor in Latin America, on its way to revolutionising the digital farming world and turning its development into a new world standard in the vet industry.
SyrinJector’s strategy focuses on creating market awareness for the new standard of digital vaccination traceability system, as well as creating the first global information system (‘big data’) of its kind.
Cohen provides futher insight: “A merger in the field of pharma – giant pharma companies are expected to show great interest in SyrinJector in order to use the product to be introduced as an enabler to their product line. A merger in the field of integrators – integrators are looking for new technologies to enrich their portfolio. A merger in the retail sector – global food companies looking for effective technologies that will enable them to support and improve their logistics system by significantly reducing costs.”
Current challenges in veterinary vaccination
The global veterinary vaccination market is facing many challenges, including the growing need for vaccines to firmly maintain livestock and public health, as well as the need to create a traceable, smarter, more user- and animal-friendly and advanced process that will enable a rapid, accurate, controlled and safe process. This trend is not limited to farms and the farmer level but goes far higher in the sales chain to consumers demanding more quality assurance for the food they are buying.
Dr Moshe Chaimovitz, former national chief veterinarian of Israel, is both an enthusiastic investor and a member of the company’s advisory board. Chaimovitz explains: “Today, animal vaccination procedures are carried out manually by mechanical syringes, by hand, needles are not replaced in time and all the paperwork involved in the process passes through manually (if at all) – without any possibility of digital control, traceability and monitoring, just as farmers did 100 years ago and more.”
Another problem arises when the human factor enters the picture, which may leave room for errors such as inconsistencies, inaccuracies, animal injury (meat damage as a result) resulting from over-use of a needle that has not been replaced on time (which also causes anxiety to the animal), and efficiency that is impaired according to the skill level of the vaccinator. As the number of animals to be vaccinated increases, so too does the chance of human error and mistreatment become greater.
The existing vaccination process places great strain on farm owners and veterinarians, who cannot ensure that the vaccines given are identical and consistent for all farm animals. In addition, farmers or vaccinators are at risk of being harmed by existing injection methods and may even be accidentally self-stabbed in the process.
“The existing process actually affects the quality and effectiveness of the vaccine; it does not allow a bio-secure (transferring disease) operation and leads to results that are not optimal in the field, while generating a lot of waste, possible pollution and high costs,” continues Chaimovitz.
The vaccine solutions currently available in the veterinary market and the mass vaccination procedure have a number of major disadvantages, including but not limited to the inability to trace, manage and monitor vaccinations performed, wrong/faulty injection, R/T animal ID reading, R/T needle replacement and disinfection between injections and more, as well as the inability to collect and analyse the relevant information in this market through big data and online monitoring applications.
The SyrinJector solution
SyrinJector has developed a solution that meets the huge market need for realising efficient and advanced digital farming, and is also applicable in the veterinary field as well. The tool is a complete digital solution based on a computerised injection syringe – an innovative, handy, automatic injecting syringe that is connected to the internet as an IoT device, and enables R/T and offline monitoring and control of the livestock‘s vaccination process.
The company has developed a comprehensive solution to enable control of the injection and make it digitally traceable and controlled in quantity, quality and safety. SyrinJector‘s product is expected to penetrate the market in the third quarter of 2019 and offer a range of digital capabilities, including information management, full automation and high accuracy. The product offers for the first time a safe and smart traceable solution for the next generation of farmers.
So how does the system work? The uniquely developed syringe is based on three main subsystems. First is the electronic syringe, which serves as an IoT syringe. The syringe was built as an electronic handy injection device with self-propulsion systems, as well as various sensing elements to ensure that the injection process is performed accurately (including false injection in a level of single animal), fast and safely.
Second is the application within any smart mobile phone with local Wi-Fi communication of up to 50 electronic syringes in parallel and a database in the cloud which includes all vaccination data per animal and is available to farmers, veterinarians, insurance companies etc. via a secured username and password.
The third component is the management system, which extracts the data (from every single injection) obtained by the syringe. The system displays all relevant data, statistics, various indications and even specific alerts to the person performing the injection. The owner of the farm can access the information on their account anytime from anywhere in the world, through the company’s cloud server.
Fig. 1 briefly describes the functions and components of the system: the injection syringe is connected via a local network in a Wi-Fi interface to the built-in smartphone app. The link between the app and the syringe does not require a cellular system or any other public or private network.
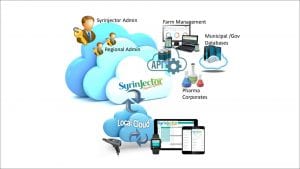
The app allows online preparation of the injection process as well as the situation in the field. From time to time and when available, the app over the smartphone uses the general data network – Wi-Fi or cellular data network, to communicate the information to the farmer account on the cloud. The cloud account allows its user to synchronise information personally and give access to authorised users.
An API (Application Programing Interface) linked to the system allow entities without an injection syringe to access information according to the administrator’s permission, for example government regulators, pharmaceutical companies, insurance companies and more.
In-depth: the business model
Of the business model in which the company is moving into, Pressler explains: “The company’s business model is based on collaborating with distributors in territories around the world, companies that distribute veterinary solutions, systems and equipment, while final potential customers for the product are the large integration corporations and farmers, who will integrate the product as a complementary device to the general digital management system. Other potential customers are pharmaceutical and retail companies, for whom the product will provide traceability – unique information previously unavailable and vital statistics regarding the use of their products.”
The company’s sales will be made through distributors/resellers of livestock equipment. For each such distributor, a programme of experiments, training sessions, planning and setting exclusive modes of operation will be created, meeting sales targets.
“We are aware of the differences in each of the markets we are dealing with,” says Pressler. In accordance with enquiries and requests from distributors, the company will also examine the possibility of leasing equipment at a monthly fee without direct purchase.
“The end user demand in this market is for a comfortable, efficient, precise and high-quality injection device. This is due to the constant increase in the need to maintain and care for animal health/welfare and well being on one hand, and on the other hand due to the complexity and limited performance of the current vaccination system – mechanical injection,” says Cohen.
Disruptive innovation and market opportunity
Veterinary injection systems, such as the product developed by the company, are becoming a critical component in the success and continued existence of the livestock market. The livestock sector has been experiencing turmoil in recent years due to various health hazards such as avian influenza, swine flu, etc. As a result, leaders and many players in the livestock industry are waiting for the development of effective solutions that will enable effective protection.
The increase of digitisation is expected in view of the increasing investment of resources by countries and key players in the market, as well as the growing awareness of the health/welfare of animals in emerging markets and in developing countries. This awareness has gained momentum in several developing countries (China, Brazil, India, etc.) which experience a significant growth of higher socioeconomic population along with exporting to Western countries that demand higher levels of supervision, quality and tracability.
Exporters such as Brazil suffer from repeated product bans due to deficiencies in the quality of the food, which causes damage to the country’s export revenue. The industry creates a reality in which the economic opportunities present themselves frequently, and there is a huge demand for developments from companies such as SyrinJector.
The situation in the pharmaceutical market in terms of marketing strategy offers a tremendous business opportunity due to the difficulty of operating within a market where everyone produces similar products for the same customers. It is very expensive to maintain any differentiation whatsoever. Pharmaceutical companies invest a fortune in encouraging customer loyalty and creating customer ‘stickiness’ – with means offering things such as free syringes and a host of small gifts to find the way to the heart and wallet of the end customer.
Leaving aside the veterinary world, there is another serious trend, Pressler points out: “Traceability of food quality assurance has now become a real issue even for supermarket chains such as Target, Sainsbury’s, Walmart, Carrefour and others. Consumers push mega food store chains to get more information about the food they put on their shelves.
“Mega chains as well as food processors are under constant pressure from their client to know more about the quality and safety chain of their livestock products. The pressure starts from users who push megastores that in turn convey their quality assurance demands over to the food processors. They want more quality assurance on the food they sell to their customers. Currently, in most of the Western countries, a high-level food quality assurance is in place, yet only one major link in this chain can’t be traced: vaccination! One actually can never know for sure who, when and if at all a vaccination took place. SyrinJector’s solution is right there as low-hanging fruit for this challenge.”
Livestock processors will have to adopt and deploy technologies and applications to have better traceability – on vaccinations as well.
Talking about the competitive landscape, Cohen says: “We see few limited concepts for swine intradermal injection from HIPRA and MSD, yet SyrinJector’s development distinguishes the company from its competitors by using a system that is open for any vaccine, regardless of the type of injection and without giving exclusivity to any vaccine manufacturer.”
This fact is added to the ability to transfer data online, which produces a significant commercial advantage in the market, which is becoming more digital with each passing year.
This is an innovative response to growing global demand in a way that completely changes and disrupts the existing market. The possibility of a digital signature injection process will become a global standard, and SyrinJector will take its place as a pioneer in this field.
Looking to the future
Pressler sets out the go-to-market vision: “SyrinJector has a broad vision for the future, and it begins with the small details. The process operates in circles that are built on one another.
“The company will start its operations with the end customer – the farmer or the injection manager – focusing on creating a solution that provides ultimate control, monitoring and safety for the process. In the first circle, the company turns to a combined global marketing process (‘umbrella’) along with local activity through authorised distributors.”
Pressler continues: “The second circle contains control entities who provide service, mainly veterinary entities who require information. This circle leads to the next one – the QA food processing chain circle. This circle enables reaching places far from the farm. For example, drug companies can obtain information about the use of medications, and insurance companies and food companies can obtain digital signatures on the implementation of vaccines in a controlled and accurate manner.
“Creating awareness for the option of a digital signature injection process will enable the assimilation of a new global standard – a kind of ‘kosher’ stamp for a safer product is definitely something I dream about at night.”
The company is also aiming at a parallel process that will enable it to become a knowledge centre for vaccines – that is, to develop for the first time a centre where vaccination activity can be analysed against health problems and/or outbreaks of diseases and epidemics and integrated with various data.
Through such analysis, it is possible to suggest links between different factors and morbidity. For example, a combination of meteorological data compared to morbidity can provide information about the relationship between drought and various outbreaks.
As for what’s next on the development agenda, Cohen says: “As we are driven by the market needs, we see a huge potential in the massive uptrend in aquaculture. We are close to introducing a similar vaccination solution for the fishery sector as well.”
SyrinJector Ltd
http://syrinjector.com/contact-us/
Please note, this article will appear in issue 8 of Health Europa Quarterly, which is available to read now.
























